Which of the following electron dot notations is correct for the element bromine, Br, atomic #35?
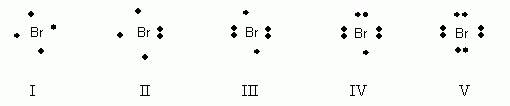
- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element aluminum, Al, atomic #13?
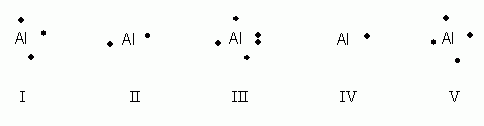
- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element argon, Ar, atomic #18?
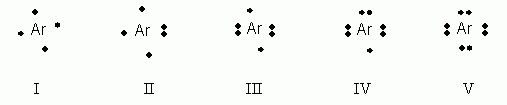
- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element arsenic, As, atomic #33?

- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element indium, In, atomic #49?
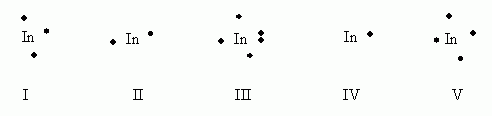
- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element calcium, Ca, atomic #20?
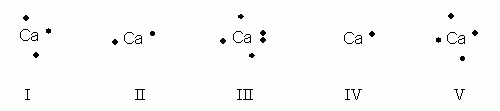
- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element carbon, C, atomic #6?
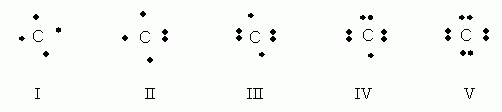
- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element oxygen, O, atomic #8?
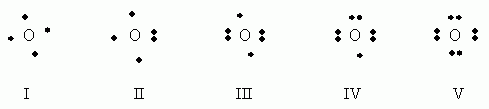
- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element phosphorus, P, atomic #15?
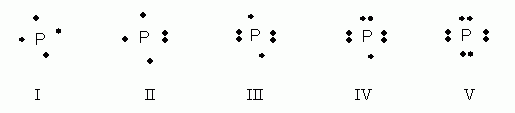
- I
- II
- III
- IV
- V
Which of the following electron dot notations is correct for the element potassium, K, atomic #19?
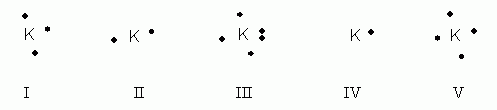
- I
- II
- III
- IV
- V
Which of the following elements has the same number of valence electrons as the element sulfur, S, atomic #16?
Which of the following elements has the same number of valence electrons as the element sodium, Na, atomic #11?
Which of the following elements has the same number of valence electrons as the element boron, B, atomic #5?
Give the correct number of valence electrons for the element fluorine, F, atomic #9.
Give the correct number of valence electrons for the element strontium, Sr, atomic #38.
Give the correct number of valence electrons for the element rubidium, Rb, atomic #37.
Give the correct number of valence electrons for the element nitrogen, N, atomic #7.
Give the correct number of valence electrons for the element krypton, Kr, atomic #36.
Give the correct number of valence electrons for the element gallium, Ga, atomic #31.
Give the correct number of valence electrons for the element silicon, Si, atomic #14.
Which of the noble gases does not have eight electrons in its outermost shell?
What element in the third series has the same number of valence electrons as bromine, Br, atomic #35?